 
Recent work on Notch ligands confirmed that the regulatory action on the extra cellular domain was crucial in human adults for self-renewal and differentiation of progenitor cells. Mutations have also been identified in mouse Notch4, which result in the absence of a regulatory action on the extra cellular domain and in mammary tumorigenesis. Missense mutations, which affect the Notch3 gene, were thought to alter the interactions with the Fringe proteins. A translocation which damages the Notch1 gene induces the expansion of undifferentiated progenitors. In mammals, several mutations in Notch genes leading to severe disorders have been reported. However, recent data show its role in cell differentiation in haematopoietic lineages, in pancreatic progenitor cell differentiation, and neural cell outgrowth. In many developing tissues, these signals inhibit differentiation. This signalling is able to regulate cell-autonomously the level of Notch partners, and to determine the fate of neighbouring cells. The intracytoplasmic domain is then translocated into the nucleus, where it associates with the transcription factor RBPJk/Su(H) to activate the expression of downstream target genes. Once the receptor is activated by a ligand from a neighbouring cell, proteolytic cleavage results in the liberation of the intracytoplasmic domain of Notch. The complexity of the Notch ligand-receptor system suggests that a high number of combined associations is necessary for multipurpose signalling. These include (a) Notch 1–4 transmembrane receptors, (b) two families of transmembrane ligands with numerous EGF repeats, Delta 1–3 and Jagged 1 and 2, and (c) three proteins modulating the ligand-receptor induced signal, termed Lunatic Fringe, Radical Fringe and Manic Fringe. In vertebrates, several molecules of the pathway have been identified. By now, it has been demonstrated to be a cell-cell signalling implicated in several developmental events: lateral cell inhibition, cell fate determination, boundary formation and patterning. This pathway includes Notch receptors, trans-membrane ligands, Delta/Jagged, and Fringe proteins. In chick and mouse embryos, it was shown to be involved in limb bud outgrowth and somitogenesis. The Notch system was first described as being responsible for neurogenesis and ectodermal specification in the fruit fly. This, and recent work on the involvement of this pathway in adults, allows us to assess the transcription level of several Notch-related genes, depending on the terminal differentiation state of the keratinocytes. 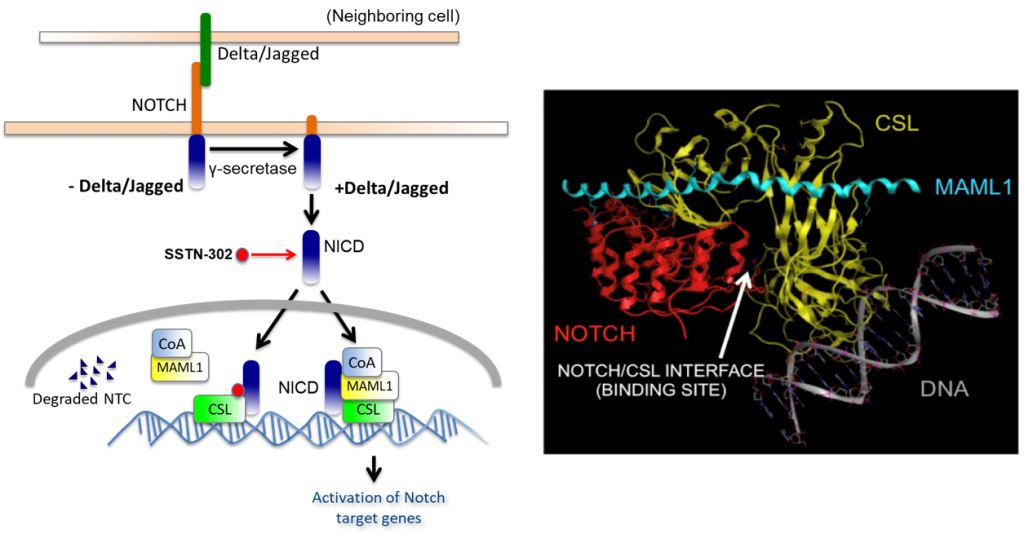
Studies on embryonic mice and rats have shown the involvement of the Notch pathway in epidermal differentiation as well as cutaneous appendage patterning. These continuous proliferative and differentiative processes result in tissue homeostasis. In mammalian epidermis, keratinocytes progress through successive phenotypic stages as they migrate from the germinative basal layer to the skin surface. The Notch signalling involved in the differentiation programme of normal adult human epidermis is altered under experimental conditions and pathologies which modify this programme. Furthermore, normal levels of transcripts were rescued in psoriatic plaques when treated by phototherapy, as well as in newly regenerated stratified epidermis following wound healing. Conversely, when keratinocytes were hyperproliferating, as in basal cell carcinoma, psoriasis, and during the first step of re-epithelialisation, expression was weak or non-existent. We show that the highest level of transcription of these genes is in the basal cell layer of non-lesional skin. Methodsīy in situ hybridisation, we investigated the in vivo expression of related genes, namely Notch 1–3, Delta 1, Jagged 1, Lunatic Fringe, Radical Fringe and Manic Fringe during keratinocyte proliferation and differentiation in humans in basal cell carcinoma, psoriasis and in wound healing experiments, compared with normal adult skin. This pathway is characterised by cell-cell interactions between transmembrane proteins and was first implicated in lateral inhibition, patterning and cell binary choices during embryogenesis. Recently, differentiation of epidermal stem cells was shown to be promoted by the Notch pathway. Epidermal homeostasis involves the monitoring of continuous proliferative and differentiative processes as keratinocytes migrate from the basal layer to the skin surface. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
